Google Ads Compliance for Aesthetic Clinics
Pay-Per-Click Compliance: Navigating Google Ads for Aesthetic Clinics

The Intricate Ecosystem of Digital Medical Advertising
The digital advertising landscape for aesthetic clinics, medical spas, and plastic surgery practices represents one of the most rigorously regulated segments within the contemporary digital economy. Operating at the complex intersection of consumer retail and strictly governed healthcare, these entities must navigate an intricate web of platform-specific algorithmic enforcements, local regulatory statutes, and federal consumer protection laws. In the current hyper-competitive environment of aesthetic medicine, Google Ads remains the preeminent acquisition channel for capturing high-intent patients actively seeking medical interventions. Unlike outbound social media marketing, which interrupts users with unsolicited content, paid search infrastructure positions medical practitioners directly in front of demographics actively querying for specific treatments or outcomes.
However, the fundamental classification of aesthetic medicine under the “Your Money or Your Life” (YMYL) paradigm necessitates aggressive policy enforcement from ad networks. Advertising platforms prioritize user safety, privacy, and financial well-being above advertiser revenue, resulting in a system where minor regulatory infractions can trigger immediate account suspensions. The primary challenge for clinic owners, practice managers, and marketing directors is no longer merely optimizing bidding algorithms or refining ad creatives; it is navigating the labyrinthine “Healthcare and medicines” policy framework. Because automated machine learning algorithms and crawling bots are responsible for the vast majority of compliance reviews, aesthetic clinics frequently experience devastating account suspensions, campaign disapprovals, and restricted reach.
These automated systems deploy deep-crawling capabilities that scan not only the text of the ad itself but the entire Destination URL. This includes the source code, metadata, global navigation menus, website footers, and dynamically loaded scripts. Consequently, a generic “set it and forget it” strategy, or a strategy overly reliant on standard agency practices, often results in rapidly depleted budgets and algorithmic penalization. In the modern landscape, where qualified Cost Per Lead (CPL) metrics range from $30 to $80 and initial competitive budgets require minimums of $1,500 per month, precision and regulatory adherence are non-negotiable. This comprehensive analysis deconstructs the mechanical, regulatory, and strategic dimensions of running Google Ads for aesthetic medicine. By examining systemic enforcement mechanisms regarding restricted drug terms, speculative treatments, personalized advertising, and cross-border jurisdictional friction, this report provides a definitive framework for achieving continuous campaign compliance while maximizing patient acquisition and return on investment.
The Restricted Drug Terms Paradigm and Context-Aware Enforcement
The most pervasive and complex compliance obstacle for aesthetic clinics is the strict governance over prescription drug terms and their active ingredients. To prevent the illicit online sale of pharmaceuticals, curb unverified medical claims, and protect consumers from unregulated substance distribution, advertising networks maintain exhaustive, continuously monitored databases of restricted drug terms.
The Monitored Substance Database
For the aesthetic sector, the restricted drug terms policy heavily impacts the promotion of neurotoxins, dermal fillers, and prescription-strength skincare regimens. The policy covers the branding, utilization, commercial sale, and distribution of these substances. The monitored list includes hundreds of active ingredients and branded pharmaceuticals. In the context of aesthetic clinics, frequently flagged terms include, but are not limited to, neurotoxins such as Botox, Botulinum Toxin, and Dysport. Furthermore, dermal fillers are aggressively monitored, including Juvederm, Restylane, and Sculptra (which utilizes poly-L-lactic acid rather than hyaluronic acid). Other aesthetic and adjunctive pharmaceuticals subject to this restriction include Latisse (bimatoprost for eyelash growth), weight management injectables (such as Ozempic, Wegovy, and Semaglutide), various hormone therapies, and anesthetics like lidocaine or buprenorphine.
When an advertiser utilizes any of these terms in their ad copy, within targeted keywords, or anywhere on their landing page without the requisite pharmacy certification, the campaign is immediately disapproved under the “Restricted medical content” or “Restricted drug terms” categorization. The enforcement mechanism is entirely automated and binary. Even if a practitioner is a highly qualified, board-certified plastic surgeon or an autonomous prescribing nurse acting entirely within their legal rights to administer these treatments in a registered clinic, the automated systems will flag the terminology. The algorithm does not possess the capability to independently verify the medical credentials or local licensing of the practitioner via the ad text; it simply detects the restricted string of text and initiates an automatic block. Furthermore, the use of trademarked products such as Botox will often trigger an additional intellectual property flag, requiring the clinic to obtain explicit permission from the actual pharmaceutical manufacturer to prove they are legally permitted to utilize the trademarked name in their advertising material.
The October 2025 Policy Update: Nuances of Non-Promotional Use
Historically, the advertising ecosystem enforced a blanket ban on the use of prescription drug terms across most global markets unless the advertiser held specific, rigorous online pharmacy or telemedicine certifications. However, recognizing the systemic friction this caused for legitimate public health initiatives, legal class-action campaigns, and educational entities, a major policy revision was enacted effective October 29, 2025.
The October 2025 update shifted the enforcement mechanism from a draconian blanket restriction to a “context-aware” approach. Under this revised framework, the non-promotional use of prescription drug terms is permitted in ad text and on landing pages globally without requiring certification. This non-promotional exception is strictly defined and applies primarily to academic publications, public health and safety campaigns, regulatory warnings, and legal notices. For example, a legal firm advertising a class-action lawsuit regarding adverse effects of a specific drug can now utilize the drug’s name in their ad text without requiring a pharmacy license.
Conversely, the promotional use of these terms—defined as any content promoting the branding, use, sale, and distribution of prescription drugs—remains strictly regulated and broadly prohibited across most international markets. Crucially, the policy regarding keyword targeting remained entirely unchanged during the October 2025 update. Bidding on a keyword that contains a restricted drug term strictly requires appropriate healthcare certification, regardless of whether the intent of the ad is promotional or purely educational. The enforcement of this updated policy ramped up over a four-to-six-week period following the October 29 launch, with algorithms designed to issue a seven-day warning to advertisers prior to any formal account suspension.
For standard aesthetic clinics and medical spas, this update offered minimal operational relief.
Because a clinic’s primary objective is the commercial promotion of services—such as selling Botox injections, hormone replacement therapies, or dermal filler administration—their use of the terminology is inherently and undeniably promotional. Therefore, if a clinic in a restricted jurisdiction mentions “Botox” on its landing page to attract paying patients, it remains in direct violation of the promotional ban.
Policy Dimension
Non-Promotional Ad Text: Prior to October 2025, this is prohibited globally without certification. Post-October 29, 2025, it is permitted globally (e.g., academic, legal, public health) without certification.
Promotional Ad Text: Prohibited globally (except US, Canada, NZ with certification) both before and after the October 2025 policy update.
Keyword Targeting: Requires explicit healthcare certification regardless of context, both before and after the October 2025 update.
Violation Penalties: Transitioning from immediate disapproval or suspension to a mandatory 7-day warning period prior to account suspension actions starting October 29, 2025.
The Certification Paradox: LegitScript and G2RS
While the global baseline strictly prohibits the promotional use of prescription drug terms, an exception exists for campaigns specifically targeting the United States, Canada, and New Zealand. In these specific jurisdictions, advertisers may utilize prescription drug terms for promotional purposes, including the branding, sale, and distribution of the drugs, provided they operate in accordance with local laws and obtain specialized healthcare certification.
LegitScript Healthcare Merchant Certification
Certification processes are not handled internally by the advertising networks but are outsourced to recognized third-party accrediting bodies, with LegitScript Healthcare Merchant Certification serving as the primary global standard. Obtaining full LegitScript certification grants medical practices the compliant authority to advertise regulated medications, such as GLP-1 agonists for weight loss, and to include brand terms in ad titles and ad copy across platforms including Google, Meta, TikTok, YouTube, LinkedIn, Netflix, and Groupon. Furthermore, since a sweeping mandate in June 2017, Visa and Mastercard require that all pharmacy merchants and telemedicine providers processing card-not-present (CNP) transactions be certified and continuously monitored by organizations like LegitScript.
The certification requirements for medical spas and aesthetic clinics are stringent. To qualify, the facility must inherently involve licensed healthcare professionals in the delivery of medical aesthetic treatments, such as injectables, dermal fillers, hormone therapy, or prescription-strength skin care. The business must actively process card-not-present transactions for these regulated services, such as facilitating virtual consultations or e-commerce pharmaceutical sales. The application process requires the submission of extensive documentation, including valid licenses for the business entity and its individual practitioners, detailed clinical policies regarding the facility’s procedures, and comprehensive ownership information.
However, a fundamental operational paradox exists for standard, brick-and-mortar aesthetic clinics seeking this certification to run local search ads. LegitScript’s healthcare certification is fundamentally engineered for online pharmacies, wholesale pharmaceutical distribution applications, price comparison websites, and robust telemedicine providers that facilitate remote prescribing. While local medical spas do employ licensed healthcare professionals who administer prescription-based treatments, the physical, in-person administration of a drug in a localized clinic setting does not typically classify the business as an “online pharmacy” or a card-not-present telemedicine provider.
Consequently, LegitScript certification is generally not applicable, nor is it granted, to standard medical spas solely for the purpose of unlocking restricted keywords like “Botox” on search engines. Because the average aesthetic clinic cannot obtain online pharmacy certification, they remain indefinitely locked out of keyword-targeting restricted drug terms. This restriction forces a mandatory strategic pivot toward symptom-based marketing and localized organic search engine optimization to capture patient intent.
Adjunctive Certifications and Application Mechanics
Beyond LegitScript, other forms of certification dictate advertising eligibility based on the specific healthcare vertical. For instance, advertisers seeking to promote health insurance in the United States must first obtain G2RS Health Insurance Providers Certification before applying to ad networks. This multi-step verification process requires a one-time application fee based on the number of U.S. jurisdictions targeted, followed by a secured application that takes up to 14 calendar days to process. If approved, the provider must pay an annual monitoring fee to receive a unique G2RS certification, which must then be submitted to the advertising network. Entities wishing to sell Affordable Care Act (ACA) qualified health plans must provide additional proof of qualification, such as a QHP Agreement or SERFF Tracking Number, to bid on ACA-related keywords.
Once a clinic or manufacturer possesses the appropriate third-party accreditation (LegitScript, G2RS, or NABP for Microsoft Ads), they must complete the specific internal application for healthcare-related advertising. This application must be submitted at the specific child account level, rather than at the Manager Account level. The applicant must select their precise organization type (e.g., Prescription drug services provider, Pharmaceutical Manufacturer, or Addiction Services Provider), select the country of licensure, and explicitly request permissions to use prescription drug terms in ad text, keywords, or landing pages. If the campaign targets allowed locations and the domain is properly certified, the ad will receive an “Eligible (limited)” label, permitting it to run in the approved geographic zones.
Speculative and Experimental Medical Treatments
In an aggressive effort to shield consumers from unproven, potentially dangerous, or scientifically unsubstantiated procedures, digital advertising platforms stringently police what they classify as “Speculative and Experimental Medical Treatments”. For the aesthetic, functional, and regenerative medicine sectors, this policy represents one of the most severe compliance minefields, often resulting in complex, site-wide technical disapprovals that paralyze lead generation.

Defining the Prohibited Categories
The platform’s definition of speculative and experimental treatments is broad and supersedes local clinical acceptance. The explicitly prohibited categories include:
- Cellular and Gene Therapies: This encompasses stem cell therapy, non-stem cellular therapy, gene therapy, and all similar forms of regenerative medicine.
- Platelet-Rich Plasma (PRP): While extensively utilized in modern aesthetics for facial rejuvenation (colloquially known as “vampire facials”) and hair restoration protocols, PRP therapies are explicitly listed as restricted experimental treatments.
- Biohacking and DIY Genetics: Do-it-yourself genetic engineering kits and biohacking products are entirely prohibited.
The enforcement of this policy is absolute and unforgiving. Even if a treatment such as PRP is legally permitted by the regional medical board, widely accepted as safe in the aesthetic community, and supported by emerging peer-reviewed clinical data, platform policy supersedes local clinical sentiment. The algorithm categorizes these treatments as lacking broad scientific or regulatory validation.
The sole exception to this rule exists within the United States, where the promotion of FDA-licensed or approved cell or gene therapies is permitted, provided the advertising entity holds the relevant FDA license or approval to market that exact product. Furthermore, advertisements for cell or gene therapies that are exclusively educational or informational in nature—such as academic papers or tickets for medical conferences—are allowed regardless of regulatory approval status. Because PRP and autologous stem cell treatments administered at local functional medicine clinics generally do not undergo the formal FDA pharmaceutical drug approval pipeline (as they utilize the patient’s own harvested tissue), standard clinics cannot utilize this FDA exception and are barred from advertising these services.
The “Guilt by Association” Crawler Mechanism
The most challenging operational aspect of the speculative treatments policy is its technical enforcement mechanism. Disapprovals are not isolated to the specific ad group actively promoting the banned service. Instead, automated web crawlers inspect the entire Destination URL and follow links throughout the broader domain structure.
If a board-certified plastic surgeon runs a fully compliant ad campaign for a standardized procedure like “rhinoplasty” or “laser hair removal,” the bot will follow the click-through link to the landing page.
If the bot detects the term “Platelet-Rich Plasma,” “Stem Cell,” or trigger phrases like “Regenerative therapy” or “Boosting immune response” anywhere on that destination domain—including within the site’s global header navigation, the footer, a drop-down menu, or an embedded blog post link—the otherwise compliant rhinoplasty ad will be immediately disapproved under the “Speculative and Experimental Medical Treatment” policy.
This algorithmic “guilt by association” forces clinics into a difficult architectural position regarding their web infrastructure. To maintain a compliant advertising account, practitioners must physically and structurally decouple their paid search infrastructure from their primary organic website. The established industry best practice involves deploying isolated, siloed landing pages hosted on subdomains or distinct third-party platforms. These siloed pages must contain absolutely no internal links back to the main website’s PRP or stem cell pages, thereby shielding the compliant ad campaigns from the restricted organic content. If a clinic fails to isolate these pages, even ads that do not mention the banned services in their copy will be halted indefinitely.
Personalized Advertising and Privacy Constraints
The capacity to deliver highly targeted, personalized advertisements is the core value proposition of programmatic digital marketing. However, healthcare is universally classified as a “Sensitive Interest Category,” triggering severe limitations on how aesthetic clinics can track, target, and retarget potential patients. These restrictions are engineered to prevent the exploitation of personal hardships, to protect user privacy, and to ensure that digital ads do not inadvertently imply knowledge of a user’s sensitive medical condition or physical insecurities.
The Prohibition of Consumer Health Remarketing
Advertising platforms strictly prohibit the use of personalized advertising features for content related to physical or mental health conditions, treatments for chronic conditions, intimate bodily functions, and invasive medical procedures. This policy explicitly lists “cosmetic surgery, surgical procedures, or injections” as restricted categories.
For an aesthetic clinic, this classification translates to a comprehensive ban on “Advertiser-Curated Audiences”. Specifically, clinics are prohibited from utilizing:
- Remarketing and Retargeting Lists: Clinics cannot track users who visited a specific service page (e.g., a “lip filler” or “acne scar” page) and serve them display ads later as they browse the broader internet.
- Customer Match: Uploading proprietary lists of past patients (utilizing emails, phone numbers, or physical addresses) to re-engage them or build lookalike models is strictly prohibited for consumer health applications.
- Lookalike Segments & Audience Expansion: Algorithmic targeting that uses a sensitive health seed list to find similar users is banned. It is worth noting that for non-sensitive campaigns, Lookalike segments began transitioning toward a “suggestion mode” in Demand Gen campaigns starting in March 2026, dropping the previous 100-user minimum seed list requirement and allowing AI to optimize beyond rigid similarity thresholds. However, because health is a sensitive category, aesthetic clinics cannot leverage these advanced lookalike features.
- Custom Segments with Sensitive Assets: Custom segments that utilize sensitive creative assets or point to sensitive landing pages will not serve at all on Search campaigns, and will only serve to non-sensitive audiences or contextually within Display campaigns.
Furthermore, all personalized advertising is strictly prohibited from serving to users under the age of 18, and advertisers are forbidden from uploading customer information derived from viewers of child-directed content.
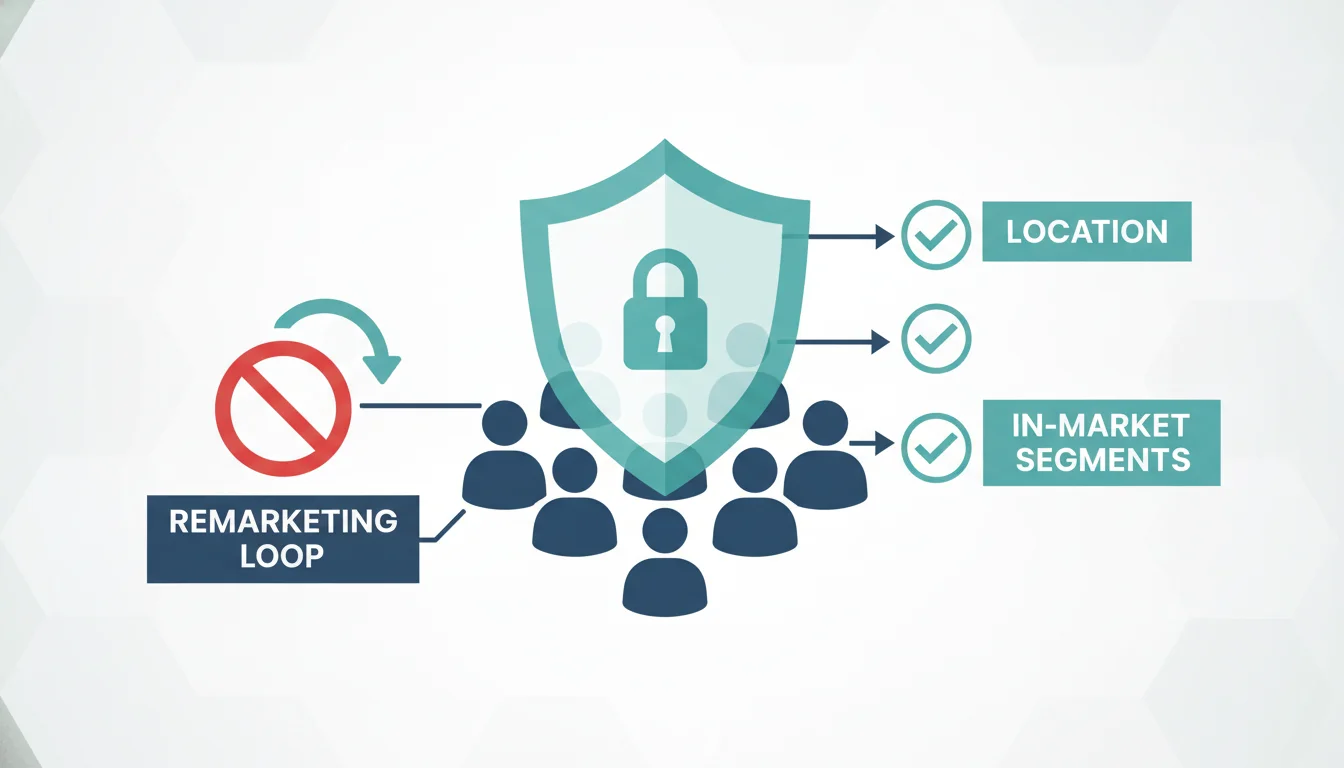
Allowed Targeting Vectors and the B2B Exception
While advertiser-curated personalized targeting is entirely prohibited, aesthetic clinics must pivot to “Predefined Audiences,” which are permitted because the advertising network automatically scrubs them of sensitive user signals. Clinics can safely utilize contextual targeting, geographic radius targeting, broad demographic targeting (age and gender), and predefined “In-Market Segments”. Utilizing in-market audiences such as “Cosmetic Procedures,” “Skincare Treatments,” or “Anti-Aging” allows clinics to layer relevance over their search campaigns without violating strict privacy constraints.
A critical nuance to the personalized advertising policy emerged via an update in July 2025. Recognizing that Business-to-Business (B2B) healthcare marketing differs fundamentally from consumer health targeting, the platform clarified that sensitive health interest restrictions do not apply to campaigns targeting licensed healthcare professionals (HCPs) in their professional capacity.
This update successfully restored the functionality of Remarketing and Customer Match lists for B2B brands, such as medical device manufacturers or pharmaceutical distributors advertising directly to clinical audiences. A physician-focused brand can now utilize restricted drug terms and personalized targeting to reach clinicians, provided they pass a newly instituted certification process specifically for restricted drug term personalization. However, this B2B exception offers zero utility to aesthetic clinics or medical spas, as their primary target audience consists entirely of direct-to-consumer (DTC) retail patients, meaning their campaigns remain highly restricted.
| Targeting Feature | Status for Aesthetic Clinics | Description / Limitation |
|---|---|---|
| Site Remarketing | Prohibited | Cannot retarget users based on previous site visits or specific actions. |
| Customer Match | Prohibited | Cannot upload patient email/phone lists for targeting or lookalikes. |
| In-Market Segments | Permitted | Can target predefined segments like “Cosmetic Procedures” or “Skincare”. |
| Contextual Targeting | Permitted | Ads match the content of the webpage being viewed, not the user’s history. |
| B2B HCP Targeting | Exempt | Remarketing allowed if targeting licensed healthcare professionals (requires cert). |
Unverified Claims, Clickbait, and Visual Evidence Policies
Beyond the aggressive filtering of drug names and experimental procedures, aesthetic advertising is heavily scrutinized for the visual and textual claims made within the ad creative and the subsequent landing page. The alignment of platform policy with federal enforcement bodies, such as the Federal Trade Commission (FTC) in the United States, mandates an exceptionally high burden of clinical substantiation.
Unverified Medical Claims and Absolute Guarantees
Platform algorithms aggressively monitor ad copy for exaggerated claims, misleading guarantees, and language that implies a “miracle cure”. In the aesthetic sector, the promise of specific physiological alterations must be tempered with clinical reality and appropriate legal disclaimers. The FTC rigorously enforces advertising standards, taking legal action against deceptive or unsubstantiated health-enhancing claims (such as the April 2026 enforcement action against TruHeight). Advertising networks design their policies to mirror this regulatory hostility toward hyperbole.
The use of absolute guarantees in ad copy is considered high-risk and is routinely banned. Clinics must strictly avoid terms such as “100% Success Rate,” “Guaranteed permanent fat loss,” “Erase wrinkles instantly,” or promises of instant results without medical backing. Furthermore, in jurisdictions like India, promising a “cure” or “100% results” is a criminal offense under the Magic Remedies Act, and the Consumer Protection Act (CPA) treats medical professionals as traders, holding them highly accountable for service claims while simultaneously restricting their ability to advertise via commissions or profit-sharing.
Compliant copywriting requires a transition from making guarantees to offering education and professional consultation. For example, instead of publishing “Lose 10lbs in 2 days with Ozempic,” compliant copy would state, “Explore medical weight management options. Doctor-led programs are available”. Instead of “Bald? Get your hair back forever,” the copy should read, “Hair restoration solutions for men. Book a consultation to discuss options”. Additionally, landing pages must prominently feature disclaimers such as “Results may vary,” and any discussion of pricing, hidden fees, or potential side effects must be entirely transparent and clearly stated. Ad copy also cannot be phrased in a way that assumes the viewer currently suffers from a medical issue or pre-existing physical defect.
The “Clickbait” Policy and Before-and-After Photography
Visual evidence of efficacy is the primary conversion driver in aesthetic medicine. Patients demand to see the practitioner’s previous work before committing to a consultation.
However, advertising platforms strictly enforce a “Before and After Images Policy,” which is often categorized under Clickbait ads or the promotion of Unrealistic Expectations.
This policy unequivocally prohibits the use of comparison photos in digital advertisements for cosmetic procedures, weight loss products, health treatments, and beauty services. The platform operates on the premise that such images set unrealistic expectations for the average consumer and typically involve unsubstantiated claims about guaranteed results. Even if the before-and-after photographs are 100% authentic, untouched, and represent actual clinic patients, they are still forbidden across all ad formats, including Search, Display, and Performance Max. Including these images in a Display Ad will lead to immediate disapproval and potential account suspension for policy violation. Clinics must relegate their visual portfolios exclusively to their organic websites and landing pages, relying instead on textual trust signals within the ads themselves.
The September 2025 “Mature Cosmetic Procedures” Update
Aesthetic clinics providing surgical interventions focused on intimate body parts historically faced immense difficulty advertising on search networks due to overarching “Sexual content” policies. Procedures such as female breast augmentation, Brazilian butt lifts (BBL), vaginal laser rejuvenation, buttock implants, pubic hair grooming, and penis enlargement surgery were routinely flagged by automated adult content filters. This resulted in severe reach limitations, shadowbanning, or outright disapprovals, despite being legitimate, medically appropriate procedures.
In a significant policy shift acknowledging the mainstream medical nature of these procedures and evolving social attitudes toward body modification, the platform announced a major update to its Sexual Content Policy effective September 22, 2025. The update officially removed “Mature Cosmetic Procedures” from the restricted sexual content category.
This implementation, which underwent a four-week deprecation period stretching into mid-October 2025, grants medical spas and plastic surgeons unprecedented freedom to market a wider array of procedures without the constant friction of automated sexual content disapprovals. Advertisers must still comply with local medical advertising laws and professional licensing requirements, and robust protections for minors remain strictly in place; advertisements promoting these mature procedures continue to be age-gated and are restricted from serving to children and teens.
Omnichannel Compliance: Microsoft, Meta, and TikTok
While Google commands the highest volume of search intent, a comprehensive digital strategy requires an omnichannel presence. However, compliance policies vary wildly across different networks, creating a fragmented landscape for healthcare marketers.
Microsoft Ads: Although not as prominent as Google, Microsoft Ads provides an excellent search engine marketing (SEM) alternative. However, its health policies are often more restrictive. While Google allows clinical trial recruitment in certain countries, it is entirely outlawed on Microsoft Ads. Microsoft forbids ads related to sensitive health conditions targeting consumers, heavily restricts weight loss product claims, and widely forbids prophylactic advertising. Similar to Google, Microsoft forbids direct health targeting (using language like “your condition” or “your symptoms”) and requires pharmaceutical sellers to receive LegitScript certification, though they additionally accept accreditation from the National Association Boards of Pharmacy (NABP).
Meta (Facebook and Instagram): Paid social platforms like Meta tightened their health advertising rules significantly leading into 2026. Meta escalated enforcement of health-related conversion tracking restrictions, limiting the use of lower-funnel optimization events for healthcare advertisers to protect user data. Health-related promotions require exceedingly careful wording to avoid making claims about outcomes or cures, and sensitive topics like weight loss or fertility require extreme caution. To run compliant Meta ads without tanking performance, clinics must utilize HIPAA-compliant landing pages, employ secure technical integrations with their Electronic Medical Records (EMR), and understand Meta’s 3-Tier Data Restrictions to navigate privacy laws safely.
TikTok: The proliferation of influencers discussing cosmetic enhancements has made TikTok a focal point for aesthetic marketing. Cosmetic clinics are permitted to advertise on TikTok, promoting services such as dermal fillers and microdermabrasion, provided they are locally certified, target appropriately, and strictly comply with all local laws and regulations governing medical advertising in their specific region.
Global Jurisdictional Clashes: Local Law vs. Platform Enforcement
Advertising networks enforce a global baseline of policies, but they actively modify their enforcement parameters based on the specific legal and regulatory frameworks of targeted countries. The interaction between local statutory law and internal corporate risk mitigation strategies creates immense cross-border friction.
Brazil: ANVISA and CFM Regulations
Brazil represents a highly complex and contradictory regulatory environment for aesthetic marketing. The medical device and cosmetics market is overseen by ANVISA (The Brazilian Health Regulatory Agency), which mandates strict marketing authorizations (such as RDC 907/2024) and pre-marketing approvals for aesthetic products. To market cosmetics, companies must hold a valid AFE (Company Operation Authorization).
Digital platform policies for Brazil are exceptionally strict. Over-the-counter (OTC) pharmaceutical creatives are entirely prohibited in Brazil, and advertisements promoting prescription drug services are explicitly banned across the board.
Furthermore, local medical regulations clash violently with platform policies. In September 2023, the Brazilian Federal Council of Medicine (CFM) published Resolution No. 2,336/2023, modernizing medical marketing to meet the demands of the sector. Crucially, this resolution legally authorized Brazilian physicians to use “before-and-after” images to demonstrate the results of aesthetic techniques and procedures, provided they include necessary medical information on indications, evolution, and complications. The resolution also mandates that advertising include the physician’s CRM registration number and specialty. However, because digital ad networks enforce their global “Before and After Images” (Clickbait) policy uniformly, Brazilian clinics utilizing paid search cannot leverage this new legal freedom. The platform rules override local permissive laws, forcing clinics to maintain image compliance to avoid immediate suspension, creating a scenario where a legally permitted action results in an algorithmic ban.
South Korea and Other Regional Restrictions
In South Korea, local laws dictate stringent review mechanisms for medical advertising. Digital networks enforce compliance with these laws by requiring that any permitted healthcare or OTC pharmaceutical advertisements prominently display an “ad deliberation number” directly within the creative itself. This number proves the ad has been legally vetted by local health authorities. The platform specifically mandates that this deliberation number must be displayed using a font size of 8 or larger; failure to meet this exact formatting requirement results in the creative being disapproved immediately.
In the United Kingdom and much of the European Union, the promotion of Prescription-Only Medicines (POMs) directly to the public is strictly illegal. Network policies enforce this blanket ban regardless of whether the clinic attempts to obtain certification, rendering direct-to-consumer Botox or weight-loss injection ads fundamentally impossible in these regions. Meanwhile, in Australia, local laws strictly ban testimonials that reference specific clinical symptoms or outcomes, forcing clinics to carefully curate their landing page reviews to avoid regulatory fines, even if the ad platform itself permits testimonials.
The Anatomy of a “Circumventing Systems” Violation
When aesthetic clinics face repeated disapprovals for restricted drug terms or speculative treatments, there is a profound temptation to utilize technical or linguistic workarounds. Engaging in these practices inevitably triggers the “Circumventing Systems” policy, which is the most severe violation in the digital advertising ecosystem, leading to an immediate, and often permanent, account suspension.
Mechanisms of Circumvention
The system defines circumvention as utilizing ads, content, or final URLs that attempt to trick or evade the ad review processes. For aesthetic clinics, this typically manifests through several specific technical or strategic errors:
- Typographical Evasion: Attempting to bypass the restricted drug terms filter by intentionally misspelling words or using special characters (e.g., typing “B0t0x”, “Bötox”, or “Juved3rm”) is easily detected by advanced natural language processing (NLP) models. This is viewed as malicious intent and guarantees a permanent suspension for Circumventing Systems.
- Cloaking and Dynamic Routing: Cloaking involves showing policy-compliant content to the network’s crawling bots while utilizing dynamic routing to redirect human users to a non-compliant page (e.g., a page selling unapproved stem cell therapies or restricted prescription drugs).
- Plugin and Script Interference: Inadvertent circumvention frequently occurs when legitimate web infrastructure interferes with crawling bots.
The deployment of geo-routing scripts, aggressive anti-bot security software, heat-mapping tools, A/B testing overlays, or newly added CMS extensions can alter the way a page loads for the crawler. If a plugin blocks or alters the page for certain visitors, the automated systems may interpret that as an attempt to hide content, raising a red flag for cloaking even if the intent was benign.
Account Proliferation: Attempting to bypass a suspension on one account by simply opening a new advertising account using a different email address to drive traffic to the exact same domain is explicitly prohibited. The system analyzes domain ownership, IP addresses, and login patterns, grouping the accounts and suspending them all.
Best Practices for Remediation and Appeal
Recovering from a Circumventing Systems suspension requires meticulous technical auditing. Accounts are only reinstated in compelling circumstances where the advertiser can irrefutably prove compliance, transparency, and the rectification of the underlying issue.
Advertisers must conduct a comprehensive technical audit of their website to identify unrecognized automatic redirects, remove uncertified click trackers, and conduct deep security checks to ensure the site has not been compromised by malicious hackers injecting cloaked content. The advertiser must ensure that the crawler bots have easy and unhindered access to all pages on the site.
Once the technical infrastructure is clean, the clinic must submit an appeal via the Policy Manager. When submitting the appeal, the clinic must be entirely transparent. The appeal should clearly explain the technical infrastructure, outline any previous mistakes (e.g., acknowledging the accidental deployment of a redirect script or an overzealous security plugin), and detail the exact steps taken to achieve compliance. The advertiser must ensure they only use trusted payment methods and that the account is connected to a legitimate business entity. Creating bulk accounts during the appeal process will irrevocably flag the business and destroy any chance of reinstatement. Advertisers are limited to three appeals per ad; if an ad is appealed unsuccessfully three times, the automated system locks, and the advertiser must navigate customer support to file any further disputes.
Architectural Strategy: Symptom-Based PPC, SEO, and Tracking
Given the overwhelming volume of restrictions—banned pharmaceutical brand names, prohibited experimental treatments, banned visual evidence, and gutted remarketing capabilities—generic campaign strategies result in rapid budget depletion. Success in aesthetic lead generation requires “artisan” keyword sculpting, rigorous landing page compliance, and a symbiotic relationship between paid search and organic search engine optimization (SEO).
The Shift to Symptom-Based Keyword Sculpting
Because standard aesthetic clinics cannot attain the LegitScript pharmacy certification required to bid on restricted drug terms like “Botox,” “Ozempic,” or “Juvederm,” bidding on these terms is a guaranteed path to disapproval and wasted effort. Furthermore, bidding on broad, top-of-funnel consideration queries (e.g., “how does botox work” or “best filler type”) drains advertising spend on informational researchers who are not ready to convert.
The optimal compliant strategy involves pivoting entirely to “Symptom-Based” and “Outcome-Based” search queries. By targeting phrases such as “reduce fine lines,” “fix sagging jowls,” “restore skin glow,” or “tear trough correction,” the clinic bypasses the Restricted Drug Terms algorithm entirely. This strategy captures a highly educated, high-intent patient demographic actively seeking a solution to a specific insecurity.
- Restricted Brand: Example queries include “Botox near me” or “Juvederm cost.” The compliance risk is extreme and requires certification. Clinics should avoid these in Paid Search and capture them exclusively via Local SEO.
- Foundational: Example queries include “Aesthetic clinic” or “Med spa London.” The compliance risk is low. Use these as baseline broad coverage to define the business.
- Symptom/Outcome: Example queries include “Fix sagging jowls” or “Reduce fine lines.” The compliance risk is low. Clinics should allocate the majority of their budget here as it captures solution-seeking patients.
- Treatment Type: Example queries include “Lip filler,” “Microneedling,” or “Laser hair removal.” The compliance risk is low to medium. Use exact and phrase match to capture bottom-of-funnel intent while monitoring for restricted claims.
To prevent the algorithm from loosely matching the ad to restricted terms and triggering a disapproval, campaigns must heavily utilize exact match and phrase match types, combined with an exhaustive negative keyword list. Broad match bidding in this sector frequently results in budget hemorrhage due to unqualified or non-compliant algorithmic matching.
SEO and Video-to-Blog Synergy
Because clinics cannot bid on lucrative terms like “Botox,” they must dominate these keywords organically. A comprehensive SEO strategy is mandatory to offset PPC restrictions.
Local SEO is the foundation, requiring optimization of the Google Business Profile, aggressive citation building, and patient review management to ensure the practice appears in local map packs when patients search for care nearby. For on-page SEO, clinics must optimize their service pages using natural language processing (NLP) analysis to maximize topical comprehensiveness and semantic relevance, establishing the “Experience, Expertise, Authoritativeness, and Trustworthiness” (E-E-A-T) that algorithms demand in the YMYL space.
Advanced clinics are deploying “Video-to-Blog” content production strategies to capture these restricted terms. Instead of writing generic blog posts, a medical professional or subject matter expert is recorded speaking to an outline. This video is embedded into a 2,000+ word optimized blog post. This approach builds profound trust, increases time-on-site (a key SEO signal), and provides unique “information gain” that search engines increasingly value over generic, AI-generated text, allowing the clinic to rank for restricted pharmaceutical terms organically where paid ads fail. Technical SEO is also required to ensure search engines can optimally crawl the site and maximize the flow of link equity from high-authority digital PR backlinks.
Landing Page Architecture and HIPAA-Compliant Tracking
Attracting a compliant click is only the initial hurdle; the Destination URL must successfully convert the patient without triggering a post-click compliance audit. Sending paid traffic to a generic clinic homepage is highly inefficient and heightens the risk of algorithmic flagging if the homepage links to restricted organic content like PRP or Stem Cells.
Aesthetic clinics must deploy isolated, high-converting landing pages tailored to the specific symptom-based ad group. The landing page copy must focus entirely on education, avoid restricted drug brand names, and omit any absolute guarantees. Because before-and-after photos are banned in the ads themselves, these isolated landing pages must carry the burden of visual proof and trust. Landing pages should feature robust trust signals, including verified Google reviews, detailed practitioner qualifications, and high-quality imagery of the clinic environment.
Finally, to achieve visibility into campaign ROI without violating personalized advertising policies or healthcare data privacy laws (such as HIPAA in the United States), clinics cannot rely on standard platform pixels that transmit Protected Health Information (PHI) directly back to the ad network. Transmitting patient data directly violates federal law and platform policy. To circumvent this safely, advanced clinics utilize third-party, HIPAA-compliant tag managers and lead intelligence platforms. These secure systems intercept the lead, track it across all sources, and allow front-office staff to qualify the lead securely. Only after the lead is marked as qualified does the system pass anonymous, aggregated conversion data back to the ad network via custom workflows and Electronic Medical Record (EMR) integrations. This highly sophisticated architecture ensures that smart bidding strategies receive the necessary quality data to optimize from day one, entirely without breaching patient confidentiality or triggering privacy policy suspensions. By mastering this intersection of compliance, technical architecture, and strategic bidding, aesthetic practices can build a sustainable, highly profitable patient acquisition engine.


